 
According to the rules of classical physics, the electrons would eventually spiral down into the nucleus, causing the atom to collapse. 
The dense nucleus is surrounded by a swarm of tiny, negatively charged electrons.īohr, who worked for a key period in 1912 in Rutherford’s laboratory in Manchester in the UK, was worried about a few inconsistencies in this model. The vast majority of its mass is located in a positively charged central nucleus, which is 10,000 times smaller than the atom itself. Rutherford had made the startling discovery that most of the atom is empty space. In July of 1913, Danish physicist Niels Bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the Bohr atom.īohr, one of the pioneers of quantum theory, had taken the atomic model presented a few years earlier by physicist Ernest Rutherford and given it a quantum twist. John Dalton, F.R.S., 1834.The most instantly recognizable image of an atom resembles a miniature solar system with the concentric electron paths forming the planetary orbits and the nucleus at the centre like the sun. In Manchester more than 40,000 people marched in his funeral procession.įeatured image: Portrait print of Dr. Dalton consolidated his theories in his New System of Chemical Philosophy(1808–1827).Īs a Quaker, Dalton led a modest existence, although he received many honors later in life. Dalton also came to believe that the particles in different gases had different volumes and surrounds of caloric, thus explaining why a mixture of gases-as in the atmosphere-would not simply layer out but was kept in constant motion. 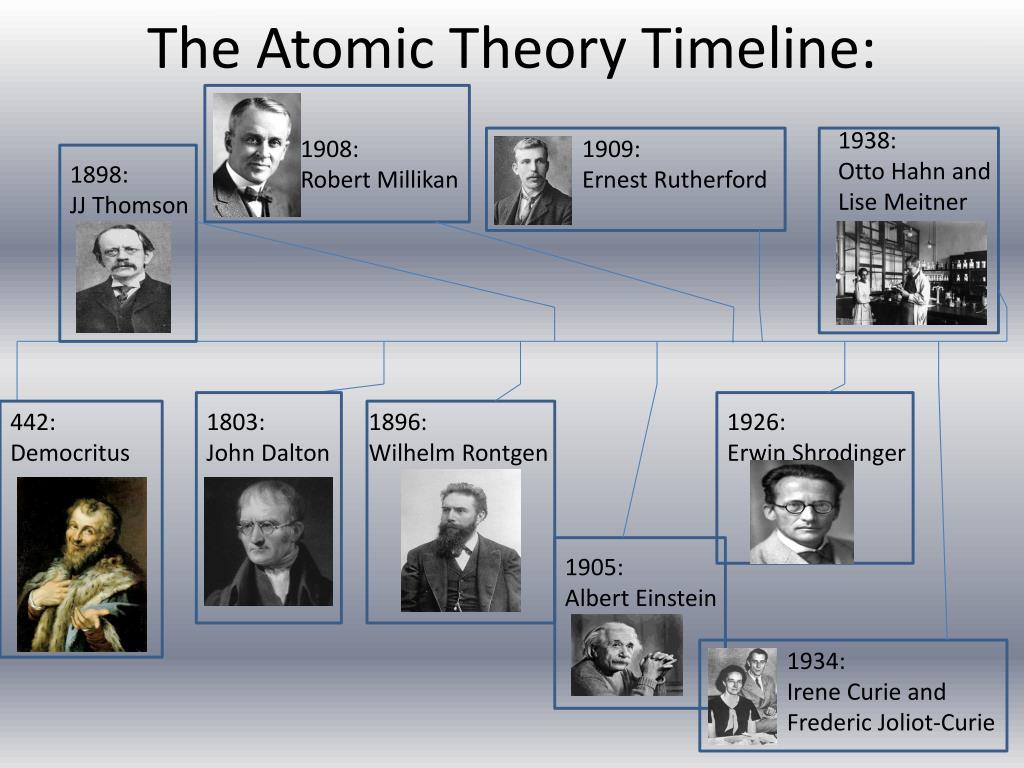
The first compound will have one atom of A and one of B the next, one atom of A and two atoms of B the next, two atoms of A and one of B and so on. If there are two elements that can combine, their combinations will occur in a set sequence. He proceeded to calculate atomic weights from percentage compositions of compounds, using an arbitrary system to determine the likely atomic structure of each compound. In explaining the law of partial pressures to skeptical chemists of the day-including Humphry Davy-Dalton claimed that the forces of repulsion thought to cause pressure acted only between atoms of the same kind and that the atoms in a mixture were indeed different in weight and “complexity.” Experiments on Atomic Weights and Structures The papers contained Dalton’s independent statement of Charles’s law (see Joseph Louis Gay-Lussac): “all elastic fluids expand the same quantity by heat.” He also clarified what he had pointed out in Meteorological Observations-that the air is not a vast chemical solvent as Antoine-Laurent Lavoisier and his followers had thought, but a mechanical system, where the pressure exerted by each gas in a mixture is independent of the pressure exerted by the other gases, and where the total pressure is the sum of the pressures of each gas.Įngraving of John Dalton by William Henry Worthington after a painting by William Allen, 1823. Theories of Atomism and the Law of Partial Pressuresĭalton arrived at his view of atomism by way of meteorology, in which he was seriously interested for a long period: he kept daily weather records from 1787 until his death, his first book was Meteorological Observations (1793), and he read a series of papers on meteorological topics before the Literary and Philosophical Society between 17. The first paper he delivered before the society was on color blindness, which afflicted him and is sometimes still called Daltonism. There he joined the Manchester Literary and Philosophical Society, which provided him with a stimulating intellectual environment and laboratory facilities. After teaching for 10 years at a Quaker boarding school in Kendal, he moved on to a teaching position in the burgeoning city of Manchester. Early Lifeĭalton (1766–1844) was born into a modest Quaker family in Cumberland, England, and for most of his life-beginning in his village school at the age of 12-earned his living as a teacher and public lecturer. He also developed methods to calculate atomic weights and structures and formulated the law of partial pressures. Science History InstituteĪlthough a schoolteacher, a meteorologist, and an expert on color blindness, John Dalton is best known for his pioneering theory of atomism. Plate 5: Elements from John Dalton’s A New System of Chemical Philosophy, 1810. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
